 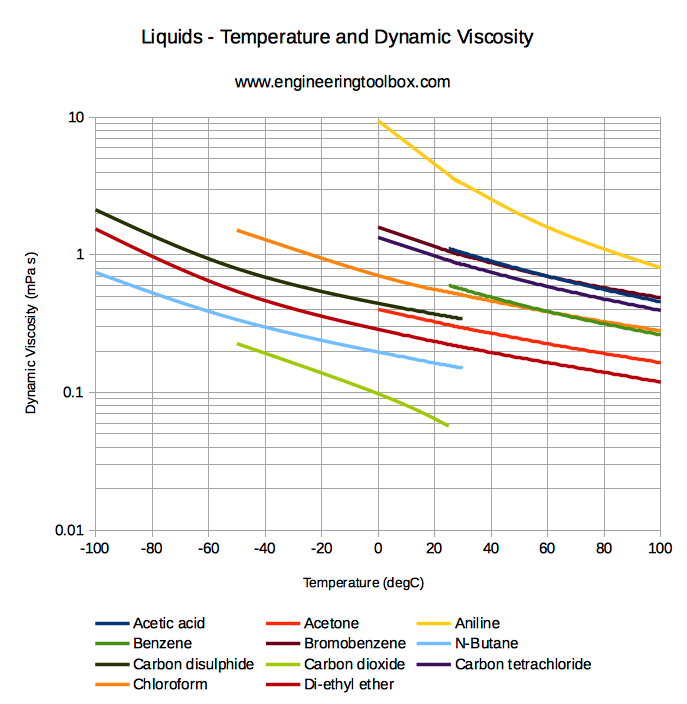
Moreover, the velocity of the spheres increases with the density of a fluid. Viscosity is measured in Pascal seconds, i.e. Δρ= difference of density of the fluid and tested sphere Velocity gradient - is the difference between the adjoining layers of liquid Note: Shearing stress: - If a direction of external force on an object is parallel to an object's plane, deformation will be along the plane and pressure felt on the object is considered shear stress. If we rest a sphere, into a liquid, we can evaluate the density by using the formula mentioned below: The density of a liquid is estimated based on a ratio of shearing stress to its velocity gradient. Below explained the symbols and terms used to calculate viscosity. The unit centistokes (cST) has its applications in various fields.Īfter knowing units of densities, it is essential to learn how to calculate densities. Poise (P) is used explicitly in ASTM standards as centipoises (cP). Since there are several types of density and each has its unit, to differentiate between dynamic viscosity and kinematic viscosity in units, students can use Poise (P) as the CGS unit of dynamic density and Stokes (St) as the CGS s unit of kinematic viscosity. Sometimes students is asked about units of viscosities. Now that students have collected some knowledge about viscosity and the difference between kinematic and dynamic viscosity, students must know about the different viscosity units. The liquid phase is probably the least well understood of all the states of matter. At ordinary temperatures, glasses are as solid as true solids. Since the process is not as well defined as true freezing, some believe that glass may still flow even after it has completely cooled, which is not the case. Molten glass is extremely viscous and approaches infinite viscosity as it solidifies. Some fats like butter or margarine are so viscous that they seem more like soft solids than like flowing liquids. Pastes, gels, emulsions, and other complex liquids are harder to summarize. Most ordinary liquids have viscosities on the order of 1 to 1,000 mPa s, while gasses have viscosities on the order of 1 to 10 μPa s. The viscosity of water at 20☌ is 1.0020 millipascal seconds (which is conveniently close to one by coincidence alone). Viscosity is first and foremost a function of the material. Simple models of molecular interactions won't work to explain this behaviour. Since liquids are normally incompressible, an increase in pressure doesn't bring the molecules significantly closer together. Viscosity is normally independent of pressure, but liquids under extreme pressure experience an increase in viscosity. That external force (F) is proportional to Shear rate (SR), Dynamic Viscosity (η), and Surface area (A). The internal resistance of a liquid flow suggests an external force applied in the movement of a liquid. Kinematic viscosity is a more fundamental property.Īpart from the difference between dynamic viscosity and kinematic viscosity, a few relations of this concept should be cleared. It is utilized when inertia and viscous force are dominant.ĭynamic force is utilized only when viscous force is dominant. This represents the ratio between shear stress to shear strain. This represents the ratio between dynamic viscosities to density. The symbol of the kinematic viscosity is V. Whereas, dynamic viscosity represents the viscous force of the liquid. It represents the inertia as well as the viscous force of the fluid. 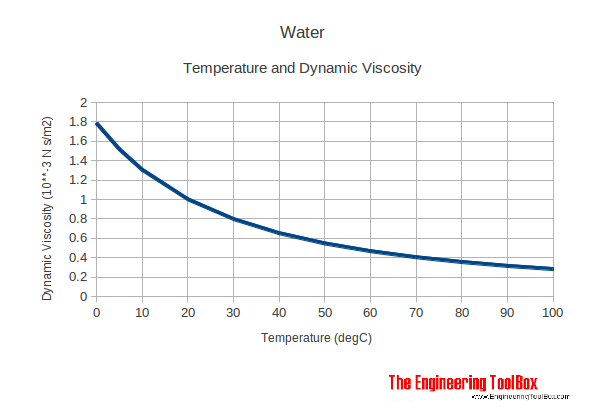
It gives more information about the force required to make the liquid flow at a specific rate. To be precise, it explains how fast the liquid is moving when a certain amount of external force is applied. This is defined as the diffusivity of momentum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
